Snapshot Day Results: pH
See Full Monitoring Results and Maps Below
Humans and aquatic creatures are both sensitive to pH.
pH is an important indicator of water quality because it tells us how acidic or basic the water is. When pH is too low or too high, it can affect whether or not organisms survive, thrive, and reproduce. For example, low pH levels allow heavy metals like copper and aluminum to dissolve in water, creating a toxic environment. Many factors can affect the pH of water, including temperature, runoff, air pollution, and algae blooms.
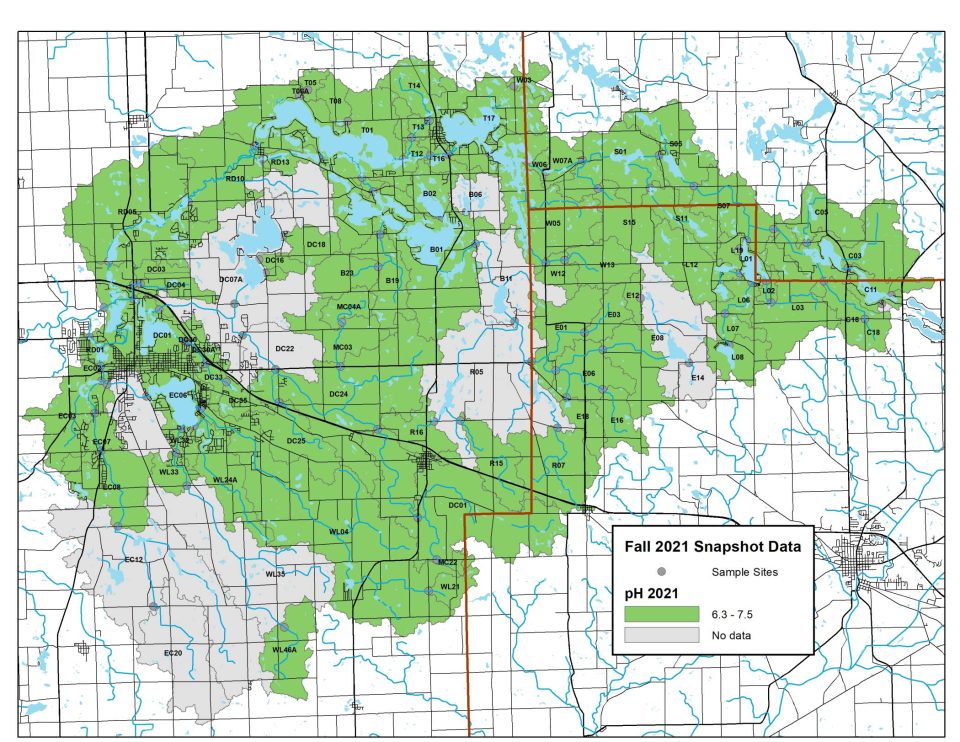
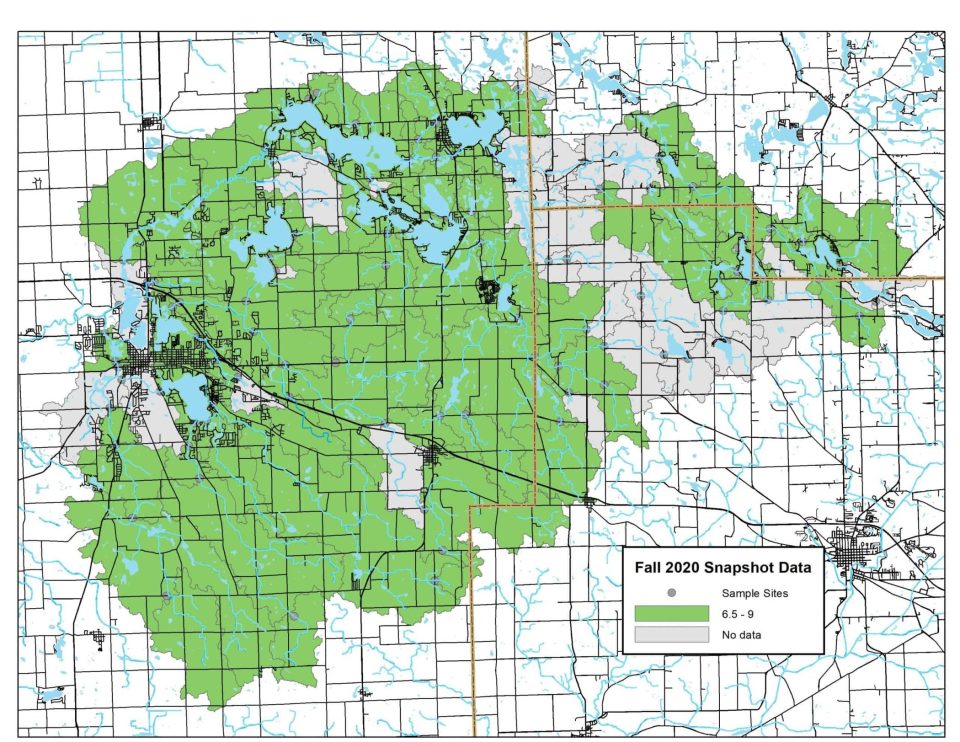
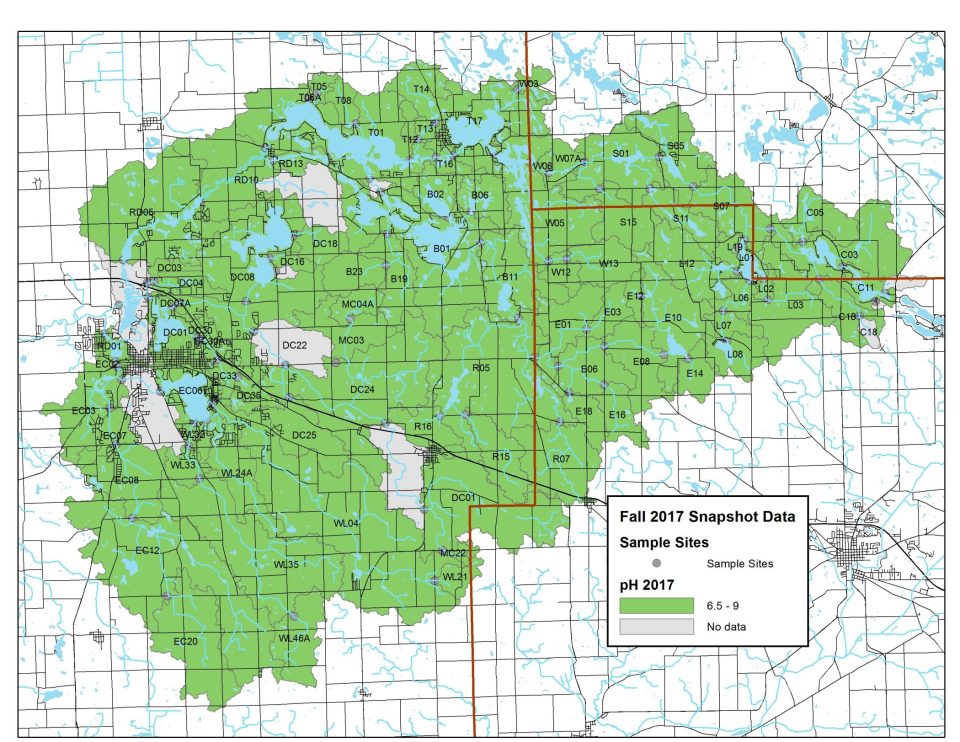
On Snapshot Water Monitoring Day, volunteers measure pH levels at sites across our watershed. Generally, pH levels continue to fall between 6.5 and 8.5, the ideal range for healthy freshwater life!
Explore full results below. Click on any map to zoom in.
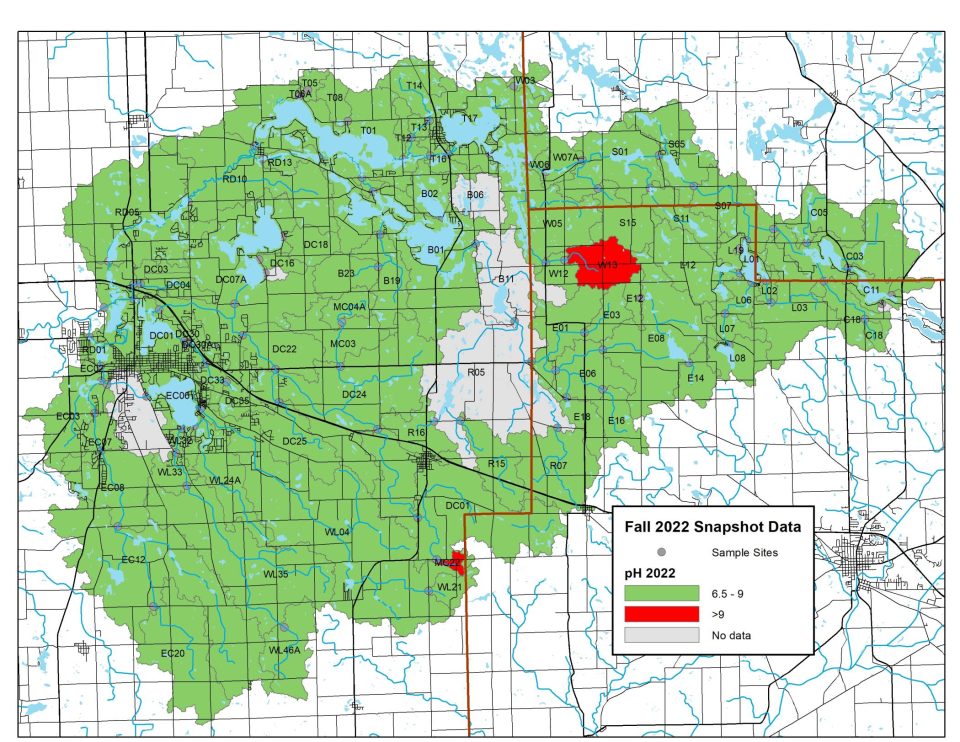
2022 pH Levels
In 2022, monitoring sites across the watershed generally showed ideal pH levels. Tests revealed two small areas of critical concern, one in the upper and one in the lower portion of the watershed.
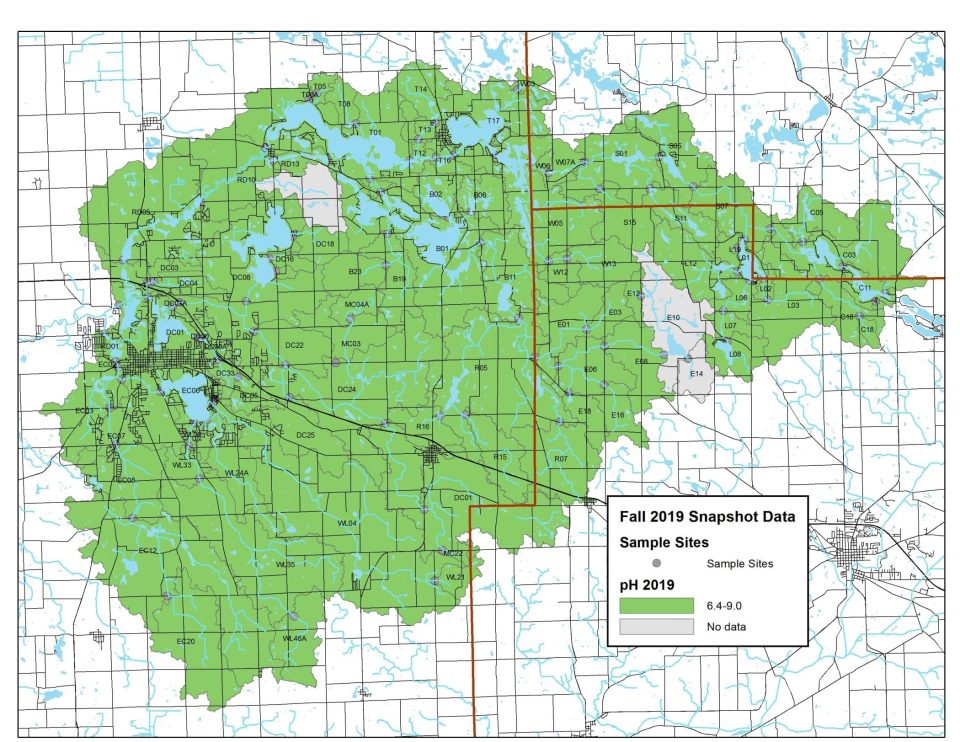
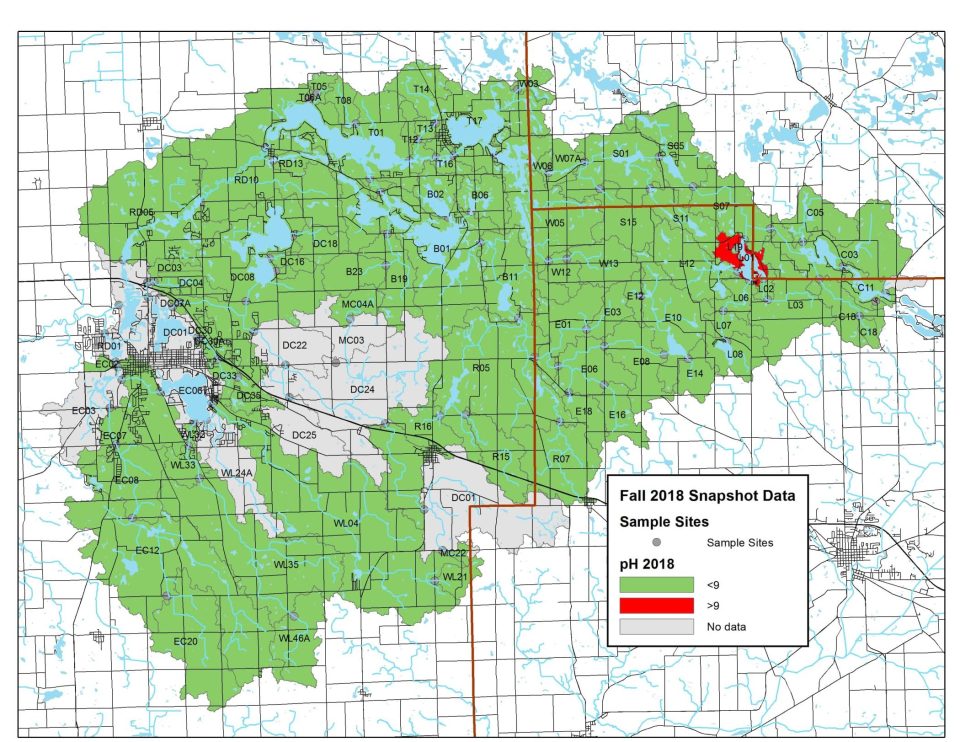
2018 pH Levels
In 2018, all monitoring sites except one showed ideal pH levels. Elevated levels were likely caused by algae blooms.